
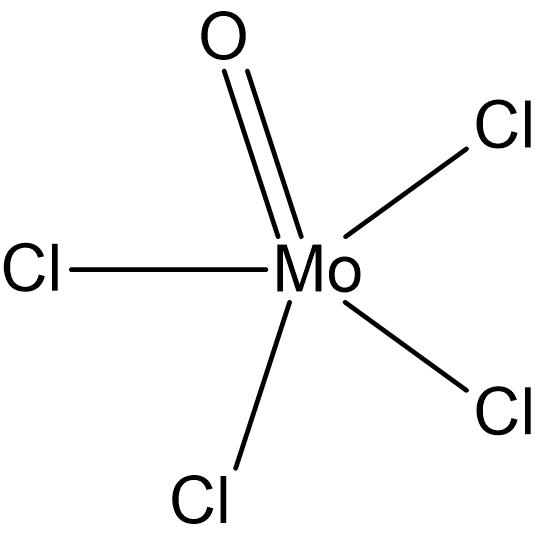
Molybdenum (VI) Tetrachloride Oxide
Properties
- We can offer items in bulk or custom. For other quantities or purities, please request a quotation.
- All prices are subject to change.
Related Products
Information about Molybdenum (VI) Tetrachloride Oxide / CAS 13814-75-0
Molybdenum Tetrachloride Oxide (MoOCl4) is a dark green solid. It is air sensitive, reacting with both oxygen and moisture. There are other known oxychlorides as well, namely Molybdenum (VI) Dioxide Dichloride (MoO2Cl2). All of these materials form a wide variety of coordination compounds. In addition to being used as a reagent, Molybdenum oxytetrachloride is also used as a catalyst.
Researchers from the Indian Institute of Petroleum in Dehradun, India were able to immobilize Molybdenum (VI) oxychloride on the surface of an ion-exchange polymer. This material was then used in the one-pot synthesis of various nitrones. The material demonstrated good catalytic activity.
Furthermore, Molybdenum (VI) Tetrachloride Oxide has also been incorporated into novel binary and tertiary catalysts. Researchers in polymer chemistry from Kyoto University in Japan prepared these novel catalysts for the so-called “living polymerization” of substituted acetylenes. The various co-catalyst mixtures they studied incorporated organometallic species including n-butyl lithium, tetra-n-butyltin, and diethyl zinc. The ternary catalysts also incorporate ethanol. These catalyst systems enabled the polymerization of monomers to occur in a controlled manner with a narrow molecular weight distribution. Such processes can be used to created block copolymers, which are used in a variety of specialized applications including adhesives, thermoplastic elastomers, and surface modification for coatings.
- Nielson, A. J., & Andersen, R. A. (1985). Tungsten and Molybdenum Tetrachloride Oxides. In S. Kirschner (Ed.), Inorganic Syntheses, Volume 23. John Wiley & Sons. https://doi.org/10.1002/9780470132548.ch41
- Larson, M. L., & Moore, F. W. (2002). Coordination Chemistry of Molybdenum Oxochlorides. Inorganic Chemistry, 5(5), 801–805. https://doi.org/10.1021/ic50039a023
- Singh, B., Jain, S. L., Khatri, P. K., & Sain, B. (2009). Nafion supported molybdenum oxychloride: Recyclable catalyst for one-pot synthesis of nitrones via direct condensation/oxidation of primary amines and aldehydes using UHP as oxidant. Green Chemistry, 11(12), 1941–1944. https://doi.org/10.1039/b914402a
- Masuda, T., Hayano, S., Iwawaki, E., & Nomura, R. (1998). Living metathesis polymerization of substituted acetylenes by MoOCl4–Et2Zn–EtOH and MoOCl4–n-BuLi systems. Journal of Molecular Catalysis A: Chemical, 133(1–2), 213–220. https://doi.org/10.1016/S1381-1169(98)00098-3
Safety
- H314Causes severe skin burns and eye damage.
- P260Do not breathe dust/fume/gas/mist/vapours/spray.
- P280Wear protective gloves/protective clothing/eye protection/face protection.
- P301+P330+P331IF SWALLOWED: Rinse mouth. Do NOT induce vomiting.
- P303+P361+P353IF ON SKIN (or hair): Remove/Take off Immediately all contaminated clothing. Rinse SKIN with water/shower.
- P304+P340IF INHALED: Remove victim to fresh air and Keep at rest in a position comfortable for breathing.
- P305+P351+P338IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses, if present and easy to do. Continue rinsing.

