
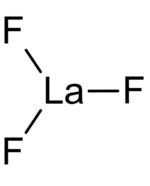
Lanthanum (III) Fluoride, anhydrous
Properties
- We can offer items in bulk or custom. For other quantities or purities, please request a quotation.
- All prices are subject to change.
Related Products
Information about Lanthanum (III) Fluoride, anhydrous / CAS 13709-38-1
Anhydrous Lanthanum Fluoride (LaF3) is a white salt used particularly in advanced optical, electronic, and metallurgical applications. For example, it is used to create fluoride glasses, anti-reflective coatings, and ion-selective electrodes.
Heavy metal fluoride glasses, such as ZBLAN, incorporate LaF3 in addition to other metal fluoride salts, including Zirconium Fluoride (ZrF4), Barium Fluoride (BaF2), Aluminum Fluoride (AlF3), and Sodium Fluoride (NaF), into their composition to attain their optical and thermal properties. Anhydrous high-purity LaF3 is crucial for successful preparation, as impurities can degrade the optical performance of the material. Trace water can form oxyfluorides, and trace transition metals causes undesirable absorption of light at various wavelengths.
In addition to its optical applications, Lanthanum trifluoride possesses exceptional mechanical properties, rendering it an ideal material for the development of anti-reflective coatings. Its high transparency across the ultraviolet, visible, and infrared light spectrums makes it a valuable component in coatings and films.
Lanthanum (III) Fluoride is commonly used in solid-state membrane electrodes for measuring fluoride ion concentrations. The sensing element consists of a Lanthanum Fluoride crystal doped with Europium (II) Fluoride (EuF₂). The Europium ions introduce vacancies into the crystal lattice, allowing fluoride ions to move through the structure by hopping between these vacant sites, which enables ionic conductivity. In the electrode assembly, the doped Lanthanum Fluoride membrane separates the sample solution from an internal reference solution. The device determines fluoride concentration by measuring the potential difference between these two solutions. Because the membrane is highly selective for fluoride ions, it provides fast and accurate measurements, particularly in acidic environments.
- Poulain, M. (2024). Heavy metal fluoride glasses: A tutorial review. APL Photonics, 9(9). https://doi.org/10.1063/5.0226668
- Lu, W., Pan, B., Miao, R., Yang, B., Yu, C., Zhang, D., Chen, D., Han, L., & Wang, Z. (2022). Dependence of Structural and Optical Performance of Lanthanum Fluoride Antireflective Films on O Impurities. Coatings 2022, Vol. 12, 12(8). https://doi.org/10.3390/coatings12081184
- Mesmer, R. E. (2002). Lanthanum fluoride electrode response in aqueous chloride media. Analytical Chemistry, 40(2), 443–444. https://doi.org/10.1021/ac60258a037
Safety
- H315Causes skin irritation.
- H319Causes serious eye irritation.
- H335May cause respiratory irritation.
- P261Avoid breathing dust/fume/gas/mist/vapours/spray.
- P302+P352IF ON SKIN: wash with plenty of soap and water.
- P305+P351+P338IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses, if present and easy to do. Continue rinsing.
- P405Store locked up.


