
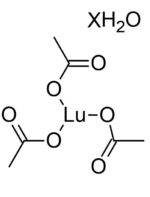
Lutetium (III) Acetate Hydrate
Properties

- We can offer items in bulk or custom. For other quantities or purities, please request a quotation.
- All prices are subject to change.
Related Products
Information about Lutetium (III) Acetate Hydrate / CAS 18779-08-3
Lutetium (III) Acetate hydrate is a colorless crystalline solid used primarily as a precursor for the preparation of Lutetium-containing advanced materials. It is water-soluble, making it suitable for sol-gel processes. It decomposes to Lutetium Oxide above 600 degrees Celsius. Advanced materials which use Lutetium include Lutetium oxyorthosilicate scintillators which are used in PET (positron emission tomography) scanners for medical imaging.
In more advanced research applications, researchers from the University of Milano-Bicocca in Italy used Lutetium Acetate in a sol-gel process to prepare Lutetium Hafnium Oxide nanopowders. The nanopowders are being studied for their use as scintillators. In the process, Lutetium and Hafnium Acetates were dissolved in water, combined with citric acid and combusted for use in their application.
Another group of researchers, from the Azcapotzalco Unit of the Metropolitan Autonomous University in Mexico City, also prepared Lutetium-based materials using Lutetium Acetate in a sol-gel process. Subsequently, the sol-gel process was annealed to produce orthovanadate powders and thin films. These materials were then utilized for the investigation of their potential applications in optoelectronic devices.
- Patil, K. C., Chandrashekhar, G. v., George, M. v., & Rao, C. N. R. (2011). Infrared spectra and thermal decompositions of metal acetates and dicarboxylates. Canadian Journal of Chemistry, 46(2), 257–265. https://doi.org/10.1139/v68-040
- González-Penguelly, B., Pérez-Sánchez, G. G., Medina-Velázquez, D. Y., Martínez-Falcón, P., & Morales-Ramírez, A. de J. (2022). Luminescence Properties and Energy Transfer of Eu3+, Bi3+ Co-Doped LuVO4 Films Modified with Pluronic F-127 Obtained by Sol–Gel. Materials 2023, Vol. 16, 16(1). https://doi.org/10.3390/ma16010146
- Lauria, A., Chiodini, N., Fasoli, M., Mihóková, E., Moretti, F., Nale, A., Nikl, M., & Vedda, A. (2011). Acetate–citrate gel combustion: a strategy for the synthesis of nanosized lutetium hafnate phosphor powders. Journal of Materials Chemistry, 21(25), 8975–8978. https://doi.org/10.1039/c1jm11553g

