
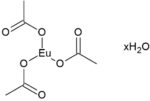
Europium (III) Acetate Hydrate
Properties
- We can offer items in bulk or custom. For other quantities or purities, please request a quotation.
- All prices are subject to change.
Related Products
Information about Europium (III) Acetate Hydrate / CAS 62667-64-5
Europium Acetate Hydrate (Eu(CH3COOO)₃·xH2O) is a white crystalline powder that is highly soluble in water, making it an ideal precursor for various synthesis methods, particularly those requiring solution-based processes. Upon heating to temperatures above 600 °C, Europium Acetate hydrate decomposes into Europium Oxide (Eu₂O₃), a highly stable material with important optical and electronic properties. The decomposition process is crucial in the preparation of europium oxide for use in advanced materials.
One of the most notable characteristics of Europium compounds, including Europium Acetate hydrate, is their ability to emit red luminescence when exposed to ultraviolet (UV) light. This property makes Europium a key element in the production of phosphors and luminescent materials, which are widely used in applications such as fluorescent lamps, TV screens, LEDs, and display technologies. The intense red emission from europium-doped phosphors is particularly sought after in the production of CRT monitors, OLED displays, and laser materials, where precise light control is needed.
Additionally, Eu(CH3COOO)₃·XH2O serves as a precursor for neutron-absorbing materials due to the neutron-capturing properties of europium, particularly in the form of europium oxide. These materials are important in nuclear reactors for controlling neutron flux and in radiation shielding applications. The versatile chemical properties of Eu(CH3COOO)₃·XH2O make it a valuable material not only in optoelectronics but also in nuclear chemistry and radiation protection technologies.
References:
- Balboul, B. A. A., & Zaki, M. I. (2011). Thermal decomposition course of Eu(CH3COO)3·4H2O and the reactivity at the gas/solid interface thus established. Journal of Analytical and Applied Pyrolysis, 92(1), 137–142. https://doi.org/10.1016/J.JAAP.2011.05.004
- Ohiienko, O. v., Moiseyenko, V. N., & Shvets, T. v. (2020). Luminescent properties of europium (III) acetate monohydrate in synthetic opal pores. Molecular Crystals and Liquid Crystals, 701(1), 72–81. https://doi.org/10.1080/15421406.2020.1732564
- Khramov, E., Belyakova, O., Murzin, V., Veligzhanin, A., Chernyshov, A., Vargaftik, M., Kozitsyna, N., & Zubavichus, Y. (2014). Investigation into the Palladium-Europium Acetate Reductive Decomposition with Synchrotron Radiation-Based X-ray Diffraction and X-ray Absorption Spectroscopy. Zeitschrift Für Anorganische Und Allgemeine Chemie, 640(12–13), 2577–2582. https://doi.org/10.1002/ZAAC.201400258
- Nakamura, K., Hasegawa, Y., Kawai, H., Yasuda, N., Tsukahara, Y., & Wada, Y. (2008). Improvement of lasing properties of europium (III) complexes by increase of emission quantum yield. Thin Solid Films, 516(9), 2376–2381. https://doi.org/10.1016/J.TSF.2007.04.092
- Perala, R. S., Putta, V. N. K., Singh, B. P., Ningthoujam, R. S., & Acharya, R. (2025). Sorption Studies of Eu3+ Ions Using YPO4 and YPO4:20% Ce Nanoparticles, Optical Properties, and in Conjunction with Instrumental Neutron Activation Analysis. ACS Omega, 10(15), 14616–14625. https://doi.org/10.1021/ACSOMEGA.4C06813\

