
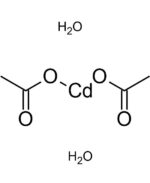
Cadmium Acetate Dihydrate
Properties
- We can offer items in bulk or custom. For other quantities or purities, please request a quotation.
- All prices are subject to change.
Related Products
Information about Cadmium Acetate Dihydrate / CAS 5743-04-4
Cadmium Acetate dihydrate is a white crystalline solid. Since it is water-soluble, it makes it a valuable precursor in the manufacture of electronics, as well as pigments.
Cadmium Acetate dihydrate is used in synthesis of Cadmium Sulfide, which is a vibrant yellow color. Thus, it has been used to make Cadmium yellow pigments. Cadmium yellows have been widely used in oil paints since their discovery in the 19th century. They are also used to color ceramics, plastics, and glasses.
Cadmium Sulfide is also used to make a variety of electronics. Notably they are used as photoresistors, devices which change electrical resistance with varying light intensity. In addition, Cadmium Sulfide is also used as component in the making of solar cells.
In recent years, Cadmium Sulfide nanoparticles have been investigated for use in nanobiotechnology, particularly for bioimaging.
- Dhatchinamurthy, L., Thirumoorthy, P., Arunraja, L., & Karthikeyan, S. (2020). Synthesis and characterization of cadmium sulfide (CdS) thin film for solar cell applications grown by dip coating method. Materials Today: Proceedings, 26, 3595–3599. https://doi.org/10.1016/J.MATPR.2019.08.219
- Ghirardello, M., Otero, V., Comelli, D., Toniolo, L., Dellasega, D., Nessi, L., Cantoni, M., Valentini, G., Nevin, A., & Melo, M. J. (2021). An investigation into the synthesis of cadmium sulfide pigments for a better understanding of their reactivity in artworks. Dyes and Pigments, 186, 108998. https://doi.org/10.1016/J.DYEPIG.2020.108998
- An, B. G., Kim, H. R., Chang, Y. W., Park, J. G., & Pyun, J. C. (2021). Photosensors-based on cadmium sulfide (CdS) nanostructures: a review. Journal of the Korean Ceramic Society 2021 58:6, 58(6), 631–644. https://doi.org/10.1007/S43207-021-00141-5
- Böer, K. W. (2011). Cadmium sulfide enhances solar cell efficiency. Energy Conversion and Management, 52(1), 426–430. https://doi.org/10.1016/J.ENCONMAN.2010.07.017
- Ghasempour, A., Dehghan, H., Ataee, M., Chen, B., Zhao, Z., Sedighi, M., Guo, X., & Shahbazi, M. A. (2023). Cadmium Sulfide Nanoparticles: Preparation, Characterization, and Biomedical Applications. Molecules, 28(9), 3857. https://doi.org/10.3390/MOLECULES28093857
Safety
- H301Toxic if swallowed.
- H312Harmful in contact with skin.
- H330Fatal if inhaled.
- H335May cause respiratory irritation.
- H340May cause genetic defects.
- H350May cause cancer.
- H372Causes damage to organs through prolonged or repeated exposure.
- H410Very toxic to aquatic life with long lasting effects.
- H302+H312+H332Harmful if swallowed, in contact with skin, or by inhalation.
- P260Do not breathe dust/fume/gas/mist/vapours/spray.
- P261Avoid breathing dust/fume/gas/mist/vapours/spray.
- P264Wash skin thoroughly after handling.
- P270Do not eat, drink or smoke when using this product.
- P271Use only outdoors or in a well-ventilated area.
- P280Wear protective gloves/protective clothing/eye protection/face protection.
- P301+P310+P330IF SWALLOWED: Immediately call a POISON CENTER or doctor/physician. Rinse mouth.
- P302+P352+P312IF ON SKIN: Wash with plenty of soap and water. Call a POISON CENTER or doctor/physician if you feel unwell.
- P304+P340IF INHALED: Remove victim to fresh air and Keep at rest in a position comfortable for breathing.
- P363Wash contaminated clothing before reuse.




